aMMP-8 First Step. First Choice
For systematic saliva screening
dentognostics GmbH is the developer and manufacturer of the aMMP-8 technology - the world's leading predictive biomarker technology for the early detection of collagen degradation processes in the body, especially in the mouth. Hidden oral inflammation causes subclinical and therefore invisible collagen degradation. Timely and personalised prevention, dental hygiene and therapy can prevent or stop this process. The aMMP-8 technology is therefore ideally suited for early detection, patient communication, awareness and monitoring of therapeutic and preventive measures because it makes the invisible visible.
Dentists
Growth. Motivation. Acceptance.
As a dentist, you rely on the organic, scalable growth of your practice. You want motivated patients who actively request treatments at your practice.
Raise your patients' awareness of the importance of your innovative treatment methods, thereby doubling their acceptance and motivation.

Oral fitness keeps your patients healthy
Preventing silent inflammation
Oral fitness influences the health of the whole body
Preventive dentistry
Preventive
dentistry
Prevention. Support. Cooperation.
With preventive dentistry, you can keep your patients' teeth healthy in the long term instead of treating them symptomatically - our aMMP-8 biomarker technology helps you to practise true prevention.
In integrated dentistry, you want to provide the best possible support for patients with chronic diseases - the aMMP-8 rapid test also reveals periodontal inflammatory processes in your patients to diabetologists and cardiologists and enables interdisciplinary collaboration.


Oral fitness keeps your patients healthy
Preventing silent inflammation
Oral fitness influences the health of the whole body
Implantology
Supplement existing diagnostics with ImplantSafe DR©

Oral fitness keeps your patients healthy
Preventing silent inflammation
Oral fitness influences the health of the whole body
Sports tooth
medicine
Sports tooth medicine
Achieve unimagined increases in performance
The link between dentistry and sport: Demonstrate your diagnostic expertise with the aMMP-8 biomarker technology and, together with your patients, achieve unimagined improvements in performance - in sport as well as in your practice.

Oral fitness keeps your patients healthy
Preventing silent inflammation
Oral fitness influences the health of the whole body
Holistic dentistry
Facilitate acceptance
Convince your patients of the benefits of alternative treatment methods with our aMMP-8 biomarker technology and facilitate the acceptance of laser and ozone therapy as well as ceramic implants.


Oral fitness keeps your patients healthy
Preventing silent inflammation
Oral fitness influences the health of the whole body
Dental
hygiene
Dental hygiene
From expert to OralFitness® coach
In der Dentalhygiene haben Sie bereits Expertise in der Prävention und Gesunderhaltung von Zähnen – und werden mit der Point-of-Care-Diagnostik zum OralFitness® Coach.

Oral fitness keeps your patients healthy
Preventing silent inflammation
Oral fitness influences the health of the whole body
Diabetology
Gynaecology
Preventive medicine
Präventions-
medicine
Personalized Precision Dentistry aMMP-8 & Vitamin D
FIRST STEP
The screening of aMMP-8 is the first and easiest step towards implementing saliva diagnostics in every practice. According to a large number of studies, aMMP-8 is currently the fastest, most precise and also the most evidence-based method for primary diagnostics to make subclinical collagen degradation measurable chairside within just 5 minutes and visible to patients in real time. In this way, we achieve intrinsic patient motivation and willingness to undergo treatment even in the early stages of a disease.
FIRST CHOICE
Unlike any other system, the patented aMMP-8 technology is the method of choice for increasing patient awareness of the need for therapeutic treatments, as saliva screening measures and visualises the collagenolytic degeneration process in real time.
dentognostics and aMMP-8 are already the first choice of...
- leading dental service organisations worldwide that use biomarker technology as a standard of care for their patients
- scientific institutions such as NASA and ESA in the field of oral health of astronauts
- renowned professional associations, such as: DGDH, DGSZM, VDDI, VDDH, DZV, GSAAM, DMS, Health Senate, Senate of the Economy
- ambitious protagonists from competitive sport from first and second division football clubs, such as the women of FC Bayern Munich, SKN St. Pölten and many more.
Universities & research institutions worldwide use aMMP-8 technology to support scientific research in the field of oral systemic health, e.g. University of Helsinki, Philipps University Marburg, University of Ulm.
In contrast to MMP-8, aMMP-8 is the biomarker of choice - the active form of matrix metalloproteinase 8. From a scientific point of view, this is the most accurate method for determining collagenolytic processes in saliva.
dentognostics is the only manufacturer in Europe to hold the coveted in vitro diagnostics authorisation for point-of-care systems and direct use in the practice.

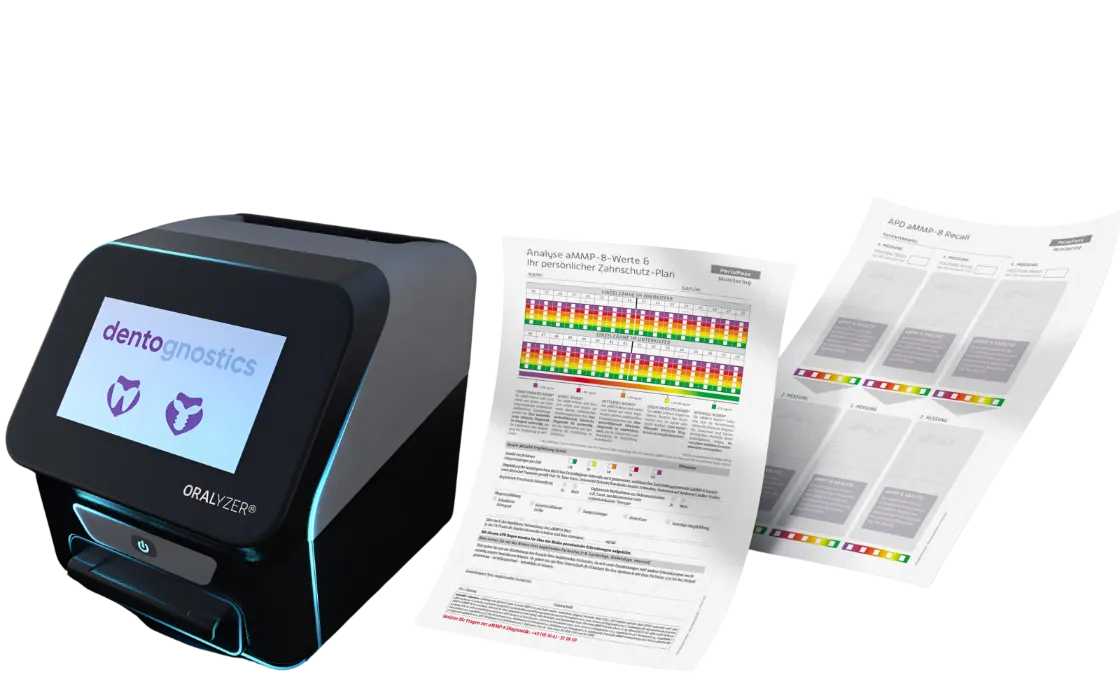

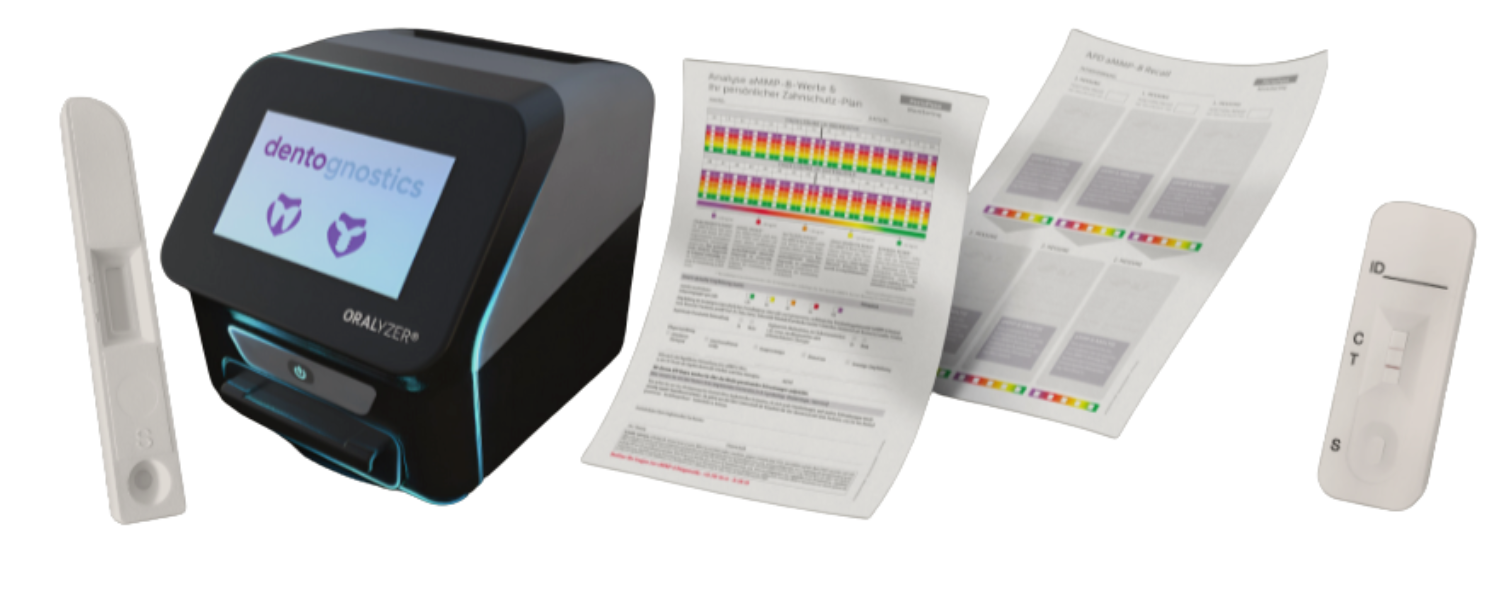
Saliva screening for your practice
Screening aMMP-8 helps you to strengthen proactive dentistry as a topic in your practice and to be a specialist in personalised prevention. Experience shows that patients who know their aMMP-8 value invest more in their oral health or implant maintenance.

Result in 5 minutes
With our modern ORALyzer® EXPERT rapid test system, you can determine the aMMP-8 and vitamin D values relevant for oral fitness quickly, safely and precisely in just 5 minutes directly at the point of care.
Personal therapy plan
Your patient receives a personalised treatment plan that you create based on their Oral Fitness Score. This will include evidence-based recommendations for your patient to take control of their oral health.
Securing the recall frequency
Once in place,
you will experience an unrivalled willingness on the part of your patients to adhere to the treatment recommendations and remain on maintenance therapy in the long term.
#1 evidence-based POC saliva diagnostics
Patented process
The aMMP-8 biomarker
(active matrix metalloproteinase-8 = collagenase 8) is the world's leading biomarker for measuring oral collagen degradation.
Making measurable
what was never measurable before. By measuring aMMP-8, active tissue degeneration processes can be recognised before they have led to irreparable damage.








